The determination of phage adsorption time is a critical step in the study of phages and their interactions with bacteria. This information can provide insights into the efficiency and specificity of phage-bacterium interactions, as well as inform the development of new phage-based therapies.
To measure phage adsorption time, a bacterial host is incubated with a phage suspension, and the number of phages attached to the host is measured at set intervals. This measurement can be done using various techniques, such as electron microscopy, fluorescence imaging, or plaque assays.
Requirements
- TSA (Some other media can be used read here)
- SM Buffer (for preparation click here)
- MgCl2
- Host bacteria
- Phage isolate
- PRE/FeSO4 solution
- Tween-80
Procedure
- Place 200μl of an appropriate SM buffer dilution of the bacterial host cell at 105 CFU/ml in a sterile Eppendorf micro-centrifuge tube.
- Add 200μl of the selected phage from the highest phage titer in the SM buffer. Mix gently to avoid causing any bubbles.
- Hold the phage-bacteria mixture at room temperature for time intervals of 1, 2, 3, 4, 5, and 10 min.
- Transfer 20μl from the phage-bacteria mixture into a sterile Eppendorf micro-centrifuge tube.
- Add 150μl of the PRE/FeSO4 solution and mix gently to avoid causing any bubbles then incubate at room temperature for 5 min.
- The activity of the virucidal agent is neutralized by adding an equal volume of 150μl of 2% (v/v) Tween-80 in SM buffer.
- After 30 s transfer the mixture into a 15 ml sterile tube and add 680μl of109 CFU/ml of 18 h bacterial host TSB culture and mix.
- Immediately add 2.5 ml of top layer agar cooled to 45°C and pour over TSA plates then incubate at 37°C for 18 h.
- Calculate the phage titers for each contact time. Contact time is given by the lowest time interval required to give the highest phage progeny for a given host at a given concentration.
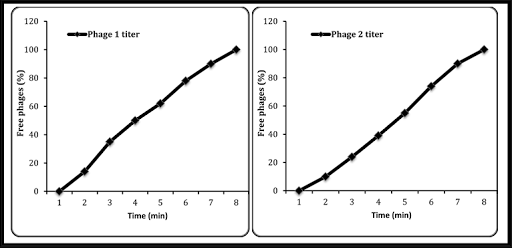
In conclusion, the determination of phage adsorption time is an important tool for understanding the behavior of phages and improving their use in the fight against bacterial infections. By providing critical information about the kinetics of phage-bacterium interactions, this technique helps researchers to better understand and optimize phage-based therapies.